Point-of-Care Ultrasound in Resource-Limited Settings
Samuel L Burleson MD, DTMH, Jonathan F Swanson MD, DTMH, Ashton E Kilgore MD, and John P Gullett MD
Department of Emergency Medicine
University of Alabama at Birmingham, Birmingham, AL, USA
Introduction
- The term “Resource-limited settings” (RLS) includes a wide range of scenarios such as hospitals and clinics in developing countries, rural areas, disaster and refugee scenarios, mobile and tactical care situations, and more.
- Acute care medicine in RLS requires adaptability and some specialized knowledge.
- Many patients in RLS lack access to diagnostic imaging.
- Even when available, traditional radiology modalities require expensive equipment, maintenance, trained staff, and specialists to interpret images.1
- Trained clinicians using point-of-care ultrasound (POCUS) may alleviate prohibitive cost, time, or travel to obtain diagnostic imaging.2
- Because confirmatory testing is often unavailable, POCUS exams may be more relevant to the patient in the RLS than in resource-rich environments.
- Conceptually, POCUS may be applied in the RLS in 3 different ways.
- Standard applications: Emergency ultrasound applications that are used routinely in emergency departments likely to be useful in any acute care setting, all of which are discussed in other chapters.3
- Advanced applications: Ultrasound applications familiar to most clinicians but requiring additional knowledge or training beyond typical POCUS curricula, based on local resource-limitations and pathology.
- Rheumatic Heart Disease
- Liver Disease
- High-risk Obstetrics
- Gastrointestinal
- Pneumonia
- Tropical disease applications: Varied and growing list of tropical infections may be diagnosed, assessed, or risk stratified by POCUS.4-6
- Hydatid cysts
- Schistosomiasis
- Dengue
- Extrapulmonary tuberculosis
- Others still being studied4
Rheumatic Heart Disease
Overview
- Rare in the developed world, rheumatic heart disease (RHD) affects 33 million people worldwide, with 320,000 deaths annually.7
- Prevalence is highest in the Pacific Islands, followed by Sub-Saharan Africa and South Asia.
- Traditional screening by history and auscultation alone is insensitive, with echocardiography as the diagnostic gold standard.8
- Echocardiography is often unavailable or impractical; handheld ultrasound has emerged as a viable screening option.9
- Echocardiography-based screening can be effectively performed by non-cardiologists.9,10
- Diagnostic criteria typically involves a combination of 2D ultrasound, color Doppler, and spectral Doppler, but this is generally only available on larger or more expensive machines.11
- Simpler criteria were developed that only require 2D and color Doppler.11 Each can be scored for risk stratification.
- Anterior mitral leaflet thickening
- >3mm measured at the tip of the anterior leaflet
- Anterior mitral leaflet thickening
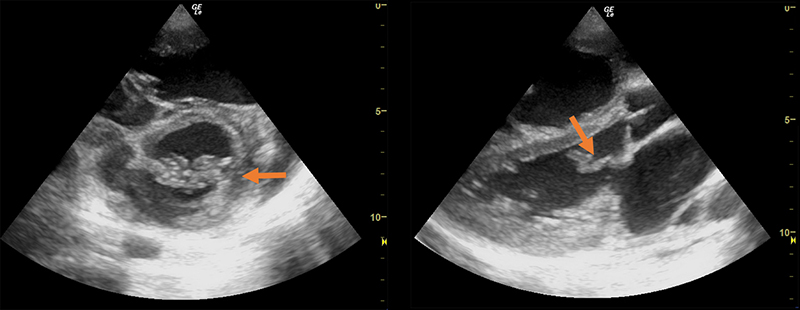
Figure 1. Parasternal short axis view (A) and long axis view (B) revealing thickened mitral valves (arrows). (Courtesy of Dr. Dan Kaminstein, Augusta University, Augusta GA, USA)
- Excessive leaf tip motion
- This refers to the "hockey stick" appearance of the anterior mitral valve (MV) leaflet seen during diastole. It will appear as though the head of the hockey stick is rotating up and down.
Figure 2. Apical 4 chamber view revealing mitral valve with abnormal hockey stick-like movement. (Courtesy of Dr. Dan Kaminstein, Augusta University, Augusta GA, USA.)
Figure 3. Parasternal long-axis view of abnormal hockey stick-like movement of thickened mitral valve.
- Mitral regurgitation (MR) jet >2 cm
- Any degree of aortic regurgitation (AR)
Figure 4. Apical four chamber view revealing both aortic regurgitation (orange jet to the right of interventricular septum in the clip) and mitral regurgitation (blue jet downwards across mitral valve).
- Irregular or focal aortic valve thickening
Figure 5A. Modified parasternal short axis view of thickened, hyperechoic aortic valve. (Courtesy of Dr. Dan Kaminstein, Augusta University, Augusta GA, USA.)
Figure 5B. Modified parasternal long axis view of thickened, hyperechoic aortic valve. (Courtesy of Dr. Dan Kaminstein, Augusta University, Augusta GA, USA.)
Performing the Scan
- Use 4 standard views as described in the cardiac chapter.
- Excessive leaf tip motion and MV anterior leaflet thickness are best assessed in parasternal long axis (PLAX) or apical 4 chamber (A4C) view.
- MR and AR jets are best viewed in A4C view, where the regurgitation jets are most parallel to the ultrasound beam, but may also be seen in PLAX.
Application
- These criteria are not yet fully externally validated or established in guidelines but may serve as useful clues for emergency clinicians, especially in endemic areas with young patients presenting with undifferentiated dyspnea, volume overload, signs of heart failure, or atrial fibrillation.
- POCUS may guide treatment, depending on local resources and care patterns.
- Secondary prophylaxis with penicillin
- Definitive cardiology care or surgical repair
Figure 6. Modified parasternal long view of advanced case of rheumatic heart disease causing severe left atrial dilation and spontaneous echo contrast (stasis of blood) in left atrium.
Liver Disease
Overview
- Liver disease accounts for approximately 2 million deaths per year worldwide, with cirrhosis and liver cancer combined accounting for an estimated 3.5% of annual deaths.12
- Many focal liver lesions are caused by tropical infections common in resource limited settings.4
- Liver abscesses (amoebic and bacterial)
- Hydatid cyst
- Schistosomiasis
- Liver ultrasound (US) may diagnose both diffuse (steatosis or cirrhosis) and focal (abscesses, masses, cysts) liver pathology.
Anatomy
- Liver Lobes - The left and right lobes are divided by the middle hepatic vein, and the caudate lobe lies posterior to the left lobe, between the inferior vena cava (IVC) posteriorly and the ligamentum venosum anteriorly.
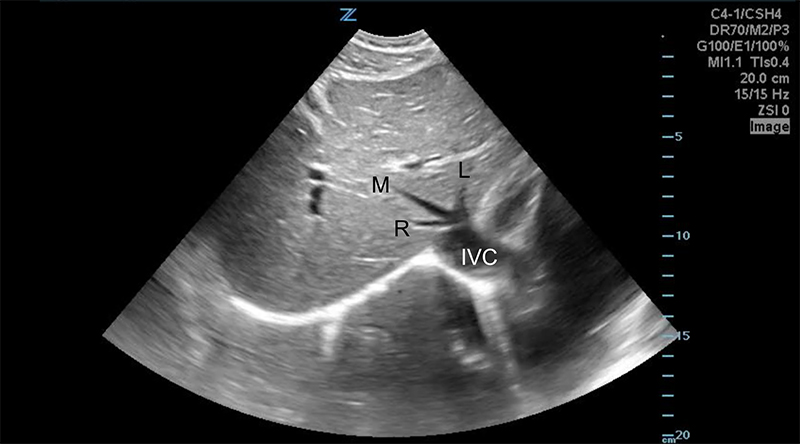
Figure 7. Transverse subxiphoid view of the liver. The middle hepatic vein divides the left and right lobes of the liver. The caudate lobe is located posterior to the left lobe. (R,M,L = Right, Middle, Left hepatic veins, respectively. IVC = Inferior Vena Cava)
- Intrahepatic structures define liver anatomy, so they should be included the sonographic image to give context and location to any focal findings. 13
- Hepatic veins
- Portal veins
- Hepatic arteries
- Bile duct branches
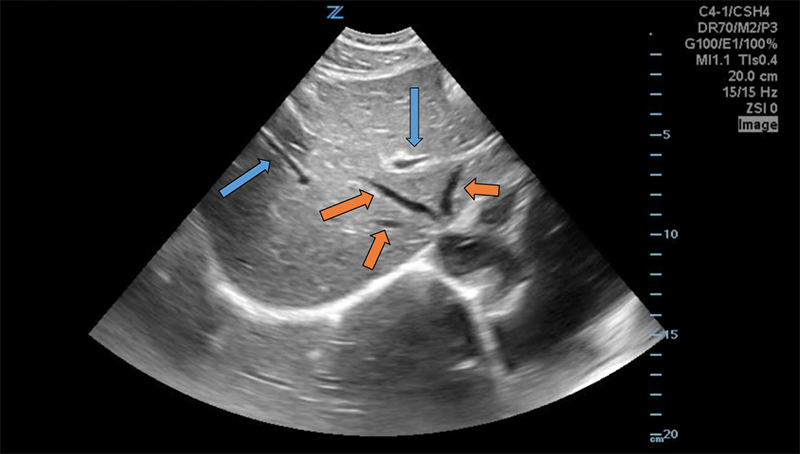
Figure 8. Transverse subxiphoid view of the liver. Hepatic veins (orange arrows) are straight with thin walls, whereas portal veins (blue arrows) have bright walls and are more tortuous.
Performing the Exam14
- Use the curvilinear probe (2-6MHz) initially, though the linear array probe (7-12MHz) may better visualize the liver margin or superficial focal lesions.
- The patient may be supine initially, but can be rolled to left lateral decubitus or the right arm extended above their head to optimize the view.
- Begin scanning in the coronal plane at the right anterior axillary line, sweeping lateral to medial through the entire liver.
- Place the probe in the axial plane (probe marker to the patient’s right) in the mid-clavicular line and fan through the entire cranio-caudal dimension of the liver.
- Rotating the probe slightly to bring it parallel to the inferior costal margin may be helpful.
- To visualize the hepatic veins, place the probe in the midline immediately subxiphoid in the axial plane. Fan cranially to the IVC/right atrium (RA) junction. Just distal to the junction, the 3 hepatic veins should be seen entering the IVC.
- To visualize the portal vein, place the probe in the midclavicular line, sagittal plane. Rotate counter-clockwise until the probe marker points to the right shoulder, then fan medially towards the aorta. The portal vein runs horizontally along the inferior surface of the right liver lobe and can be followed proximally to the junction of the superior mesenteric and splenic veins.
- The portal vein may also be located in short axis by tracing the gallbladder down its long axis to the neck, forming the “exclamation point”
- Further details can be found in the biliary chapter.
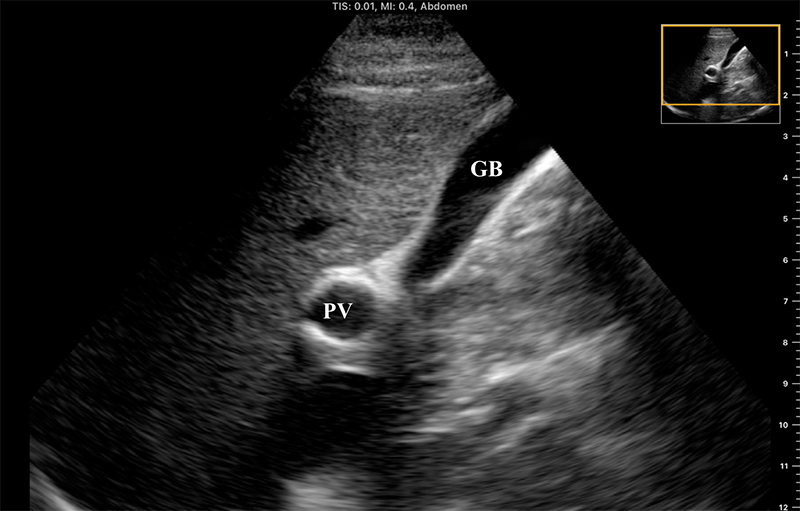
Figure 9. “Exclamation point” revealing the gallbladder (GB) in long axis with the bright-walled portal vein (PV) in short axis.
- The larger, bright-walled portal vein will often be seen with the two smaller structures of the portal triad in the same cross section, a hepatic arterial branch and common bile duct.
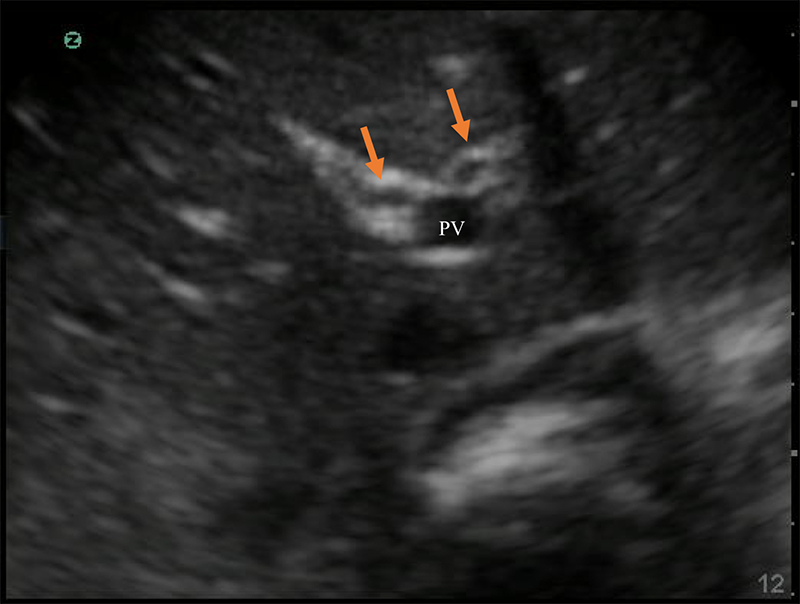
Figure 10. The portal triad seen in short axis. The portal vein (PV) is the largest, while the hepatic artery and bile duct (arrows) are much smaller and often indistinguishable without color Doppler.
Normal Findings
- Size of liver
- 12-14 cm in sagittal antero-posterior dimension at mid-clavicular line. 15
- <16 cm from posterior superior to anterior inferior.13
- Surface
- Smooth without lumps, nodules, or indentations.16
- Texture
- Fine homogenous texture of moderate echogenicity,15 brighter than the renal cortex, and darker than the spleen.14
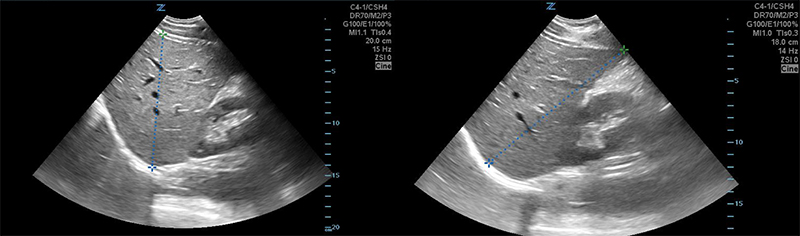
Figure 11. Normal Liver as seen from the right flank coronal view. Note the smooth surface, fine homogenous texture, and tubular vascular structures. The normal anterior-posterior diameter in the mid-clavicular line sagittal plane is between 12-14cm (A). The normal maximum dimension is from anteroinferior to posterosuperior is <16cm (B).
- Tubular structures15
- Hepatic veins are generally straight with thin or imperceptible walls.
- Portal venous branches have brighter, echogenic walls and curved or rounded courses, making it more difficult to visualize more than a short section in a single frame.
Figure 12. Normal portal vein in long axis. Note the bright hyperechoic walls of the portal vein.
- Diffuse Pathology17
- Hepatic Steatosis
- Typically increased echogenicity of liver diffusely especially when compared to kidney.
- Decreased visualization of intra-hepatic tubular structures secondary to beam attenuation.
- Finer echotexture.
- Cirrhosis
- Surface nodularity
- May be better appreciated with linear probe and has excellent positive predictive value. 16
- Parenchymal nodularity and heterogeneity of echotexture.
- Shrunken, though may have preserved caudate lobe in moderate cases.
- Surface nodularity
Figure 13. Cirrhotic liver with surrounding ascites. Note the surface nodularity of both anterior and posterior surfaces, parenchymal irregularity, shrunken liver size, and decrease in vasculature as compared to a normal liver.
Figure 14. Another example of a cirrhotic liver with surrounding ascites.
- Often associated with signs of portal hypertension.
- Ascites
- Splenomegaly
- This finding is non-specific in isolation, especially in areas where other tropical infections are endemic4
Figure 15. Splenomegaly with the spleen extending below the inferior pole of the left kidney.
- Enlargement of portal vein (>14mm)
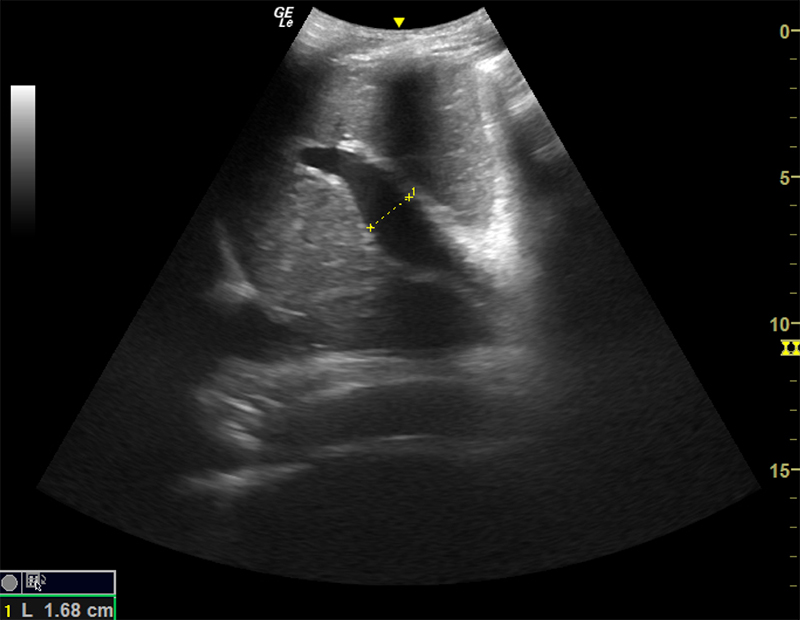
Figure 16. Dilated portal vein (>14mm) in the setting of portal hypertension.
- Reversal of portal venous flow
- Physiologic blood flow in the portal vein is from deep (the spleen and intestines) to superficial (the liver periphery), towards the probe.
- With the probe on the anterior abdominal wall, blood in the portal vein flows from central (the mesenteric and splenic veins) towards the liver and will be red when color Doppler is applied. The middle and left portal venous branches will still appear red. However, blood in the right portal venous branch will flow deep into the right lobe, appearing blue.
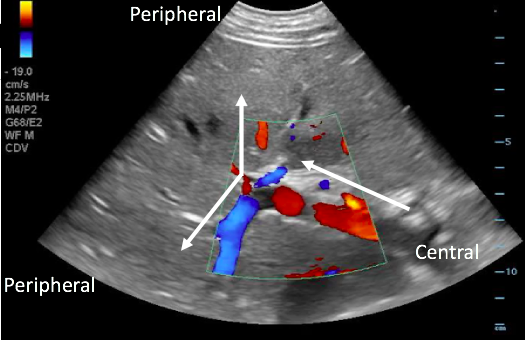
Figure 17. Color Doppler image with arrows illustrating normal portal venous blood flow, from the center towards the periphery of the liver.
- May indicate portal hypertension, as increased pressure forces blood back towards the spleen and intestines
- Blue flow (away from probe) in the portal vein on color Doppler.
- Color Doppler may serve as a useful screening tool.
- Spectral Doppler measurements may also be applied, but are often unavailable in RLS and are beyond the scope of this chapter.
- Viral Hepatitis
- Often undetectable sonographically.
- Thickening of walls of the portal triad may be visible.
- Focal Liver Lesions17
- The following is a partial list of the lesions most pertinent to the acute care provider in RLS.
- Many of these lesions may have variable sonographic presentations, and ultrasound may not provide a definitive diagnosis. Therefore, the POCUS exam should be used in conjunction with clinical findings, patient and epidemiologic risk factors, and other diagnostic resources as available.
- Most common appearances for each lesion are emphasized.
- Evaluate each lesion with Doppler color flow as this is a diagnostic criterion for several and may differentiate between vascular and cystic structures.
- Cysts
- Simple cysts are the most common liver lesion and features include:
- Thin, single wall
- Anechoic lumen
- Posterior acoustic enhancement
- No Doppler flow
- Usually small
- Simple cysts are the most common liver lesion and features include:
- The following is a partial list of the lesions most pertinent to the acute care provider in RLS.
Figure 18. Multiple simple liver cysts with thin, single walls and anechoic contents.
- Complex cysts most often due to hemorrhage, but may be related to other causes of focal lesions as below. Sonographic features of complex cysts include:
- Internal echoes
- Septations
- Thick walls
- Solid elements
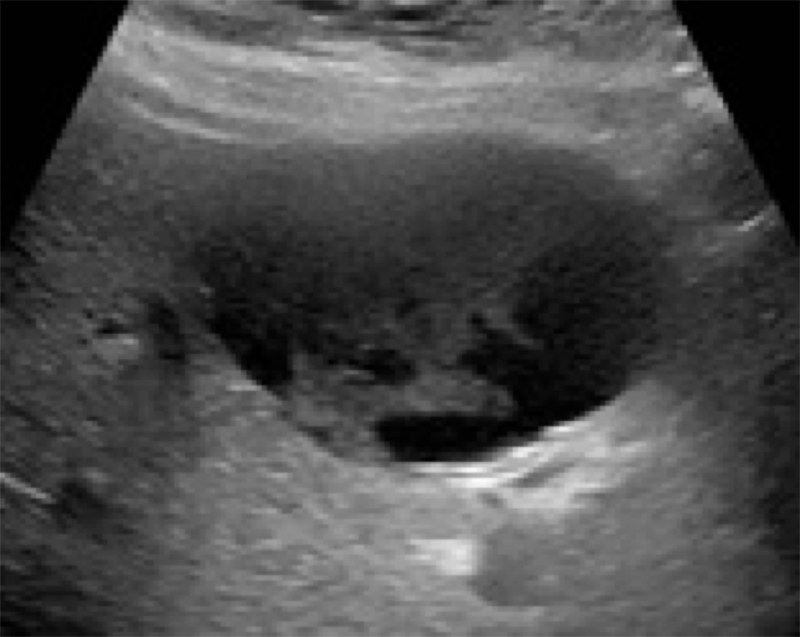
Figure 19. Hemorrhagic cyst with echogenic contents and no septations. (Liver Imaging Atlas, www.liveratlas.org. Copyright 2010, University of Washington, Image used with permission from University of Washington, accessed 4/14/21)
- Tumors
- Hemangiomas
- Most common benign tumor.
- Sponge-like structure of many small vessel walls filled with blood.
- Often asymptomatic.
- Homogenous, hyperechoic, and usually <3cm on ultrasound, with distinct and smooth margins.
- May be multiple.
- Blood flow is generally too slow to detect with Doppler flow.
- Hemangiomas
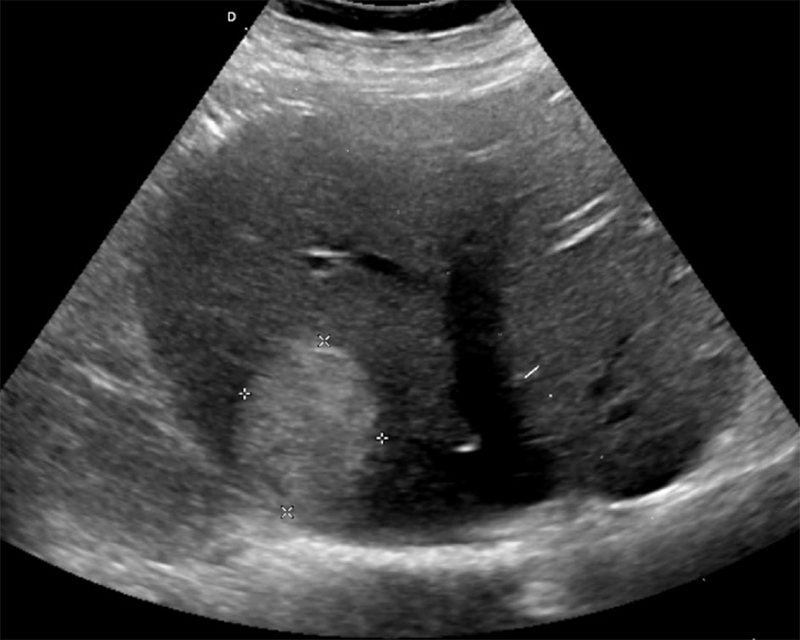
Figure 20. Homogenous echogenic lesion on gray-scale ultrasound in the right hepatic lobe posteriorly with no color Doppler flow. (Liver Imaging Atlas, www.liveratlas.org. Copyright 2010, University of Washington, Image used with permission from University of Washington, accessed 4/14/21)
- Metastases
- Most common malignant liver lesion (in North America)
- Often multiple
- May have a wide variety of sonographic appearances
- Roughly spherical and hypoechoic
- Solid and hypoechoic
- Calcification and shadowing
- Cystic or liquefied central core
- Thick septations
Figure 21. Multiple intrahepatic slightly hyperechoic masses. Note the heterogeneous “bumpy” appearance of the liver parenchyma.
- Often present as “target lesions”.
- Hyperechoic or isoechoic center with hypoechoic halo.
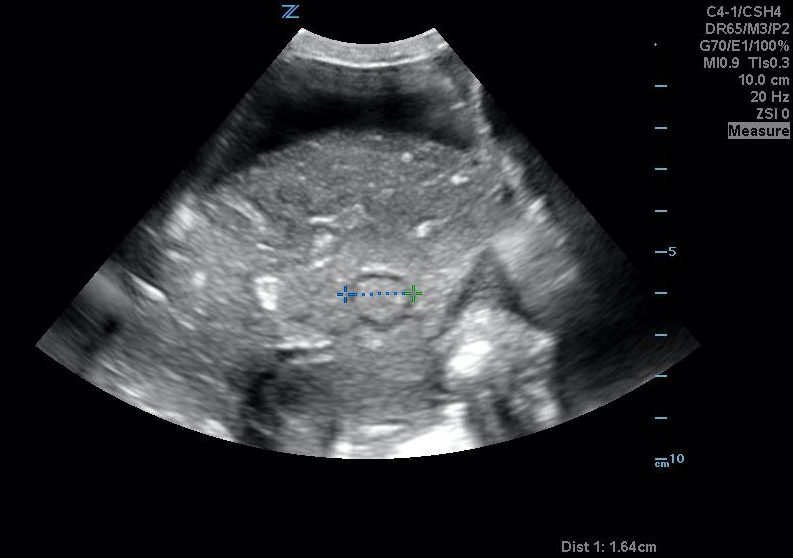
Figure 22A. Uterine metastasis seen as an intrahepatic target lesion.
Figure 22B. Uterine metastasis seen as an intrahepatic target lesion.
- Sonographic appearance cannot predict source of primary lesion.
- Hepatocellular carcinoma
- Most common primary malignancy of the liver, often secondary to alcohol abuse or chronic viral hepatitis.
- Variable sonographic appearance, including echogenicity and texture.
Figure 23. Axial view of liver demonstrating multifocal hyperechoic lesions due to hepatocellular carcinoma. Note the surrounding ascites.
- A large dominant lesion with smaller “satellite” lesions is typical, but not sensitive.
- Often hypervascular and may invade hepatic vasculature.
- Abscesses and infections
- Pyogenic abscess
- Most commonly secondary to infections in the intestines, such as appendicitis, diverticulitis, or direct extension from gallbladder infection or instrumentation.
- Bacterial etiology will vary based on geographical location and patient exposures.
- Many pyogenic liver abscesses are polymicrobial, with specific etiologies based on region and individual patient risk factors18
- In North America, coli, Klebsiella, and Streptococcal species are most common.
- Enteric gram-negative species are common causes related to biliary disease.
- Appear as complex fluid collections with mixed echogenicity, thick walls, septations, fluid levels, and posterior acoustic enhancement.
- Occasional hyperechoic gas layers may cause “dirty shadowing.”
- Often “geographic” in distribution, with irregular borders.
- Most commonly secondary to infections in the intestines, such as appendicitis, diverticulitis, or direct extension from gallbladder infection or instrumentation.
- Pyogenic abscess
Figure 24A. Large pyogenic liver abscess with heterogenous material swirling inside the lesion, and “dirty shadowing.”
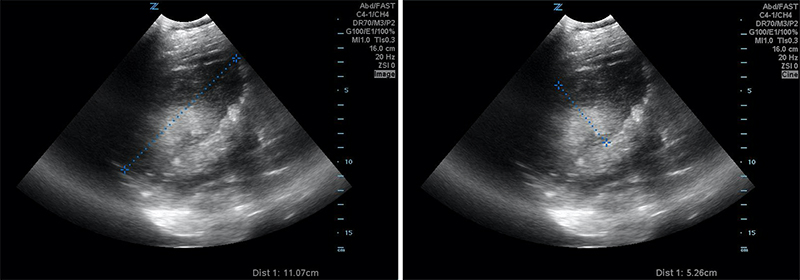
Figures 24B & 24C. Large pyogenic liver abscess with heterogenous material swirling inside the lesion, measuring >11cm in the long dimension (B) and >5cm in the shorter dimension (C).
- Amoebic abscess4
- Prevalent in sub-Saharan Africa, Indonesia, Indian subcontinent, Mexico, and parts of Central and South America.18
- Often presents 2-3 weeks after intestinal amoebic infection with fever, RUQ pain, and jaundice.
- Most often single, usually in the posterior right lobe.
- Thin or invisible wall, hypoechoic, more uniform texture.
- More common than pyogenic abscesses in the tropics.
Figure 25. Large right lobar liver amoebic liver abscess. Note the complex heterogeneity and dirty shadowing.
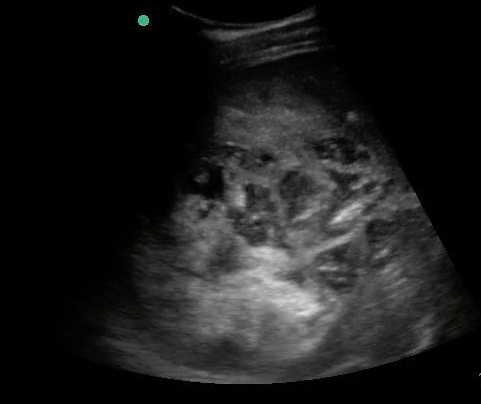
Figure 26. A still image of the same right lobar liver amoebic abscess.
Figure 27. Transverse epigastric view of a separate left liver lobar amoebic abscess in the same patient.
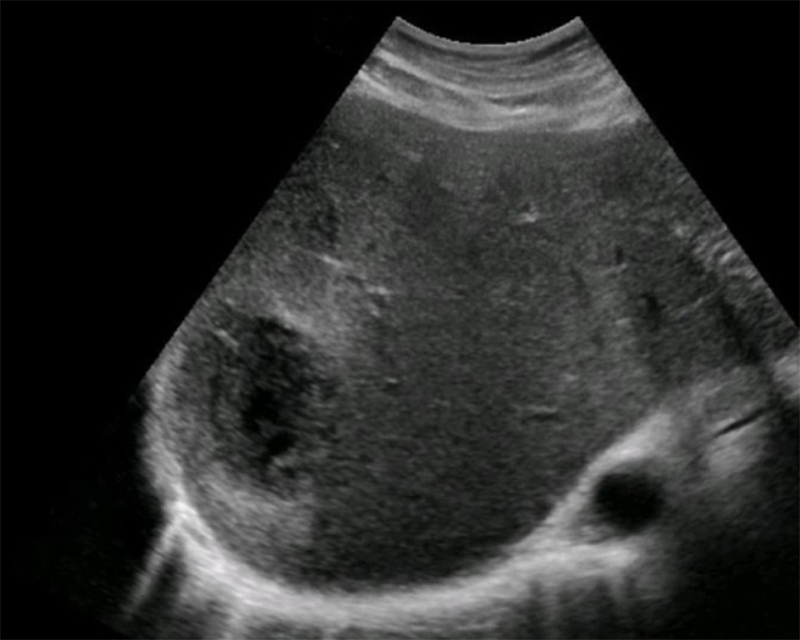
Figure 28. Well-defined hypoechoic fluid collection in the right liver lobe, with central fluid, complex periphery, and a thin wall representing an amoebic abscess. (Liver Imaging Atlas, www.liveratlas.org. Copyright 2010. Image used with permission from University of Washington. Accessed 4/14/21)
- Hydatid cyst (see section below)
Obstetrics
- Obstetric ultrasound (OB US) performed by emergency clinicians in many resource-rich environments is often limited to the confirmation of an intra-uterine pregnancy, dating, fetal heart tones, and movement for well-being.
- This paradigm is predicated on a mother having relatively close follow-up with OB/GYN, recurrent imaging, and planned delivery in a hospital.
- Maternal and fetal mortality both occur at a rate at least ten times more frequently in RLS than high income settings. 19
- Both obstetric textbooks20 and a recent large international randomized controlled trial (RCT) 21 describe a relatively simple second or third trimester OB US protocol assessing risk of maternal or fetal complication and need for referral to and delivery at advanced healthcare facilities in RLS.
- Mechanical Risk Factors for Delivery
- Fetal Presentation
- Number of Fetuses
- Placentation
- Fetal Well-Being
- Fetal Heart Tones
- Amniotic Fluid
- Fetal Biometry
- General physicians and non-physician clinicians can use and interpret OB US22,23
- Several smaller studies reveal OB US leads to changes in the management of individual patients22,24
- A large international RCT21 did not find improved patient-oriented outcomes or increased antenatal care after the systematic addition of US to routine pre-natal care in a variety of RLS.
- Despite this evidence against its systematic inclusion in routine prenatal care, OB US in the hands of a trained emergency clinician may still be beneficial in the evaluation and management of an individual pregnant patient, especially one with symptoms.
- Particularly in settings where blood products and surgical delivery are limited or absent, and detection of predictors for complicated delivery could lead to earlier referral, intervention, or resource mobilization.
- Fetal Presentation
- If the fetal cranium is in the pelvis, then the fetus is vertex.
- Mechanical Risk Factors for Delivery
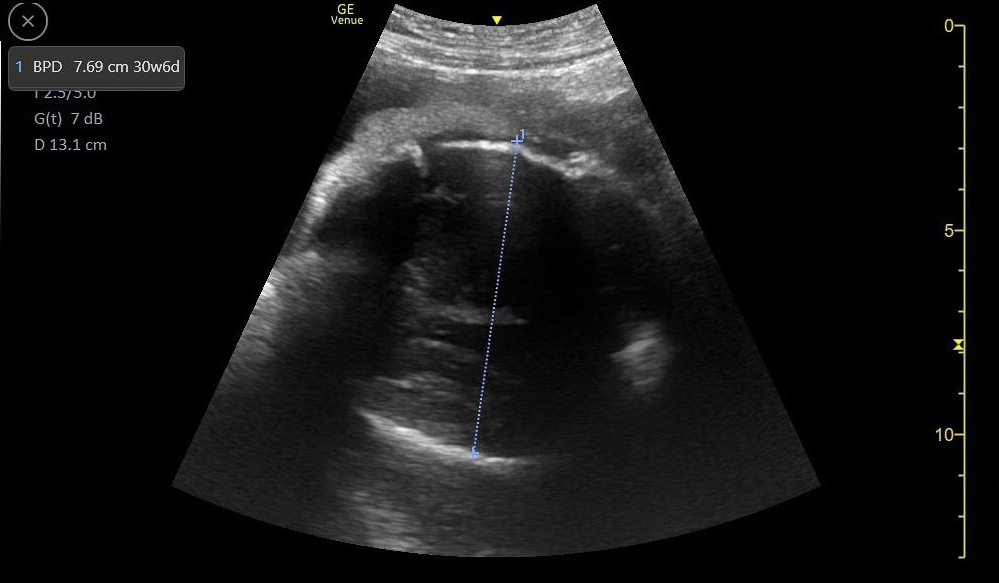
Figure 29. Transverse suprapubic view revealing the fetal cranium in the pelvis, consistent with a vertex presentation. Note the calipers measuring a bi-parietal diameter at 30 weeks and 6 days.
- Non-vertex (breech, transverse, or oblique) increases risk of difficult delivery and complication25
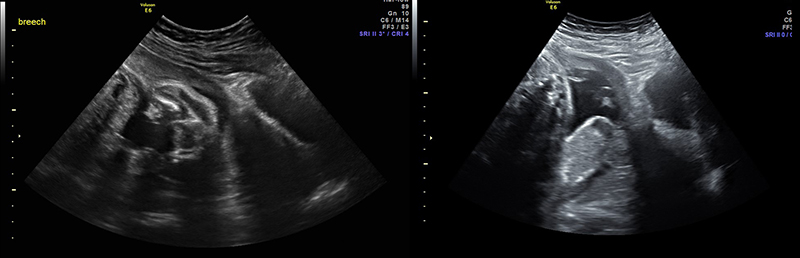
Figures 30A & B. Suprapubic views revealing no fetal cranium in the pelvis. While in some cases like this transverse view of a breech presentation (A) and this sagittal view of a transverse presentation (B), it may be difficult to exactly determine the fetal anatomy, the lack of the fetal cranium is adequate to rule out a vertex presentation and should increase the clinician’s suspicion for a more difficult delivery.
- Number of Fetuses
- Multiple pregnancies increases risk of preterm delivery, preeclampsia, abnormal labor, and intrauterine growth restriction26
- Often diagnosed by finding two fetal heads or two fetal poles in uterine cavity.
- Confirm by identifying 2 separate fetal bodies.
- Dividing membrane often seen, but not required in monochorionic-monoamniotic twins.
Figure 31. Note two separate third trimester fetal crania.
Figure 32A. Two completely separate chorionic and amniotic sacs are visible. Two different eggs were fertilized and the children will be fraternal not identical twins. Note, it can be difficult to distinguish dichorionic diamniotic from other types of twins such as diamniotic monochorionic, especially early in pregnancy.
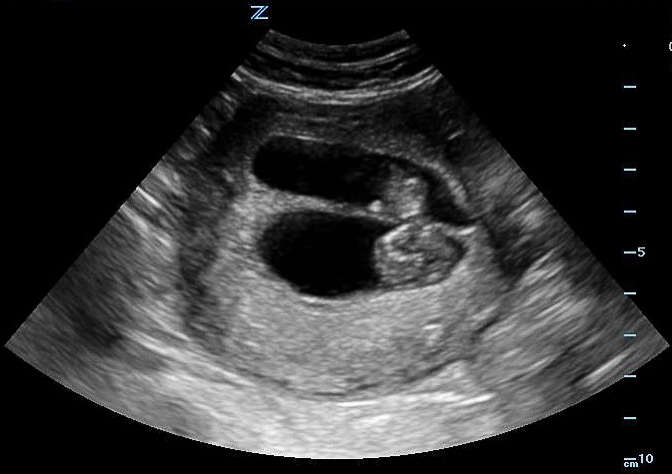
Figure 32B. Two completely separate chorionic and amniotic sacs are visible. Two different eggs were fertilized and the children will be fraternal not identical twins. Note, it can be difficult to distinguish dichorionic diamniotic from other types of twins such as diamniotic monochorionic, especially early in pregnancy.
Figure 33A. Two separate sacs are visible. In this case dichorionic diamniotic, but could also be monochorionic but not monoamniotic, in which the fetuses would appear to share the same space.
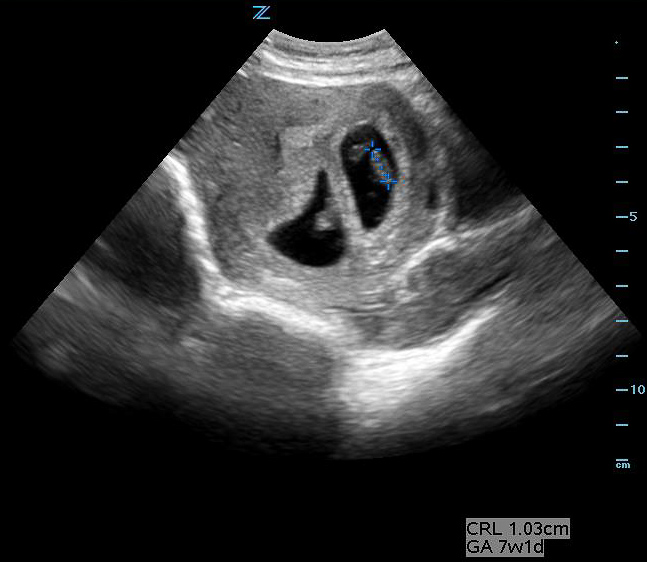
Figure 33B. Two separate sacs are visible. In this case dichorionic diamniotic, but could also be monochorionic but not monoamniotic, in which the fetuses would appear to share the same space.
Figure 34. Dichorionic diamniotic twins with multiple body parts and dividing membrane seen.
- Placentation
- Abnormal placentation such as placenta previa increases the risk of peripartum maternal hemorrhage.27
- Detecting abnormal placentation prior to delivery may allow preparation for increased hemorrhage or operative delivery and lower risk to the mother.
- Placenta previa (covering the internal cervical os) or low-lying placenta (within 2 cm but not covering the cervical os) should be confirmed by transvaginal US if available.
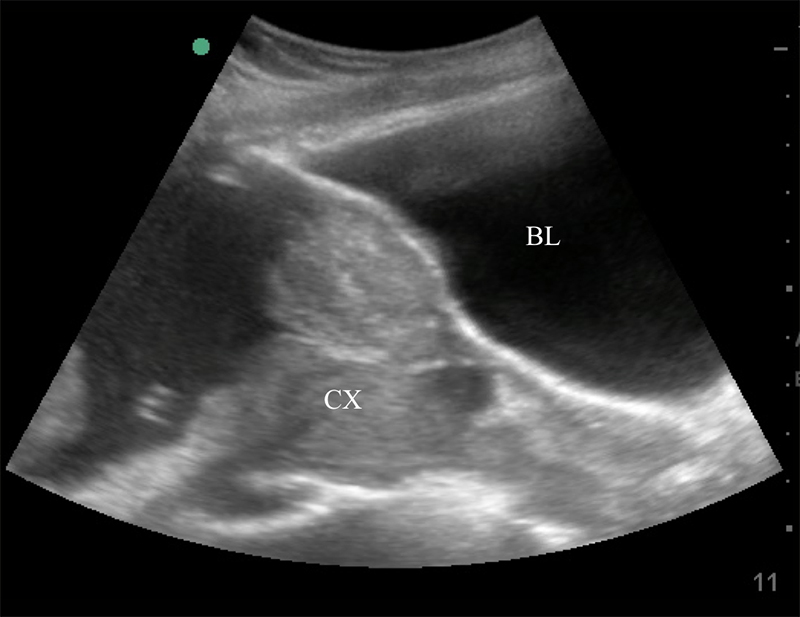
Figure 35. Sagittal transabdominal view of the pelvis revealing the anechoic urinary bladder (BL) and a normal cervix (CX) just below.
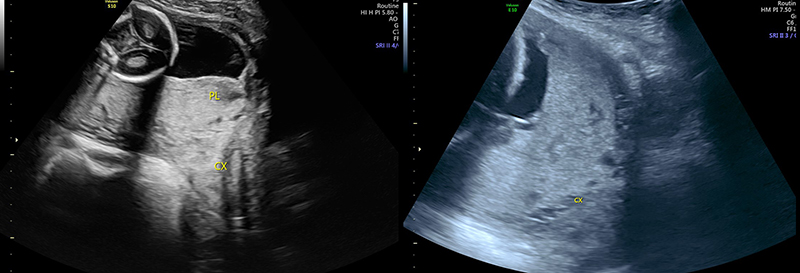
Figure 36. Sagittal transabdominal view of the pelvis similar to Figure (prior), with hyperechoic placenta (PL) covering the internal cervical os (CX).
- Fetal Heart Tones
- Same technique as in early pregnancy.
- Greatest Vertical Pocket (GVP).
- After scanning the entire uterine cavity systematically (mowing the lawn), find and measure the single deepest pocket of anechoic amniotic fluid perpendicular to the bed with no fetal parts present. The probe marker should be pointing to the patient’s head.
- Normal ranges from 2cm – 8cm
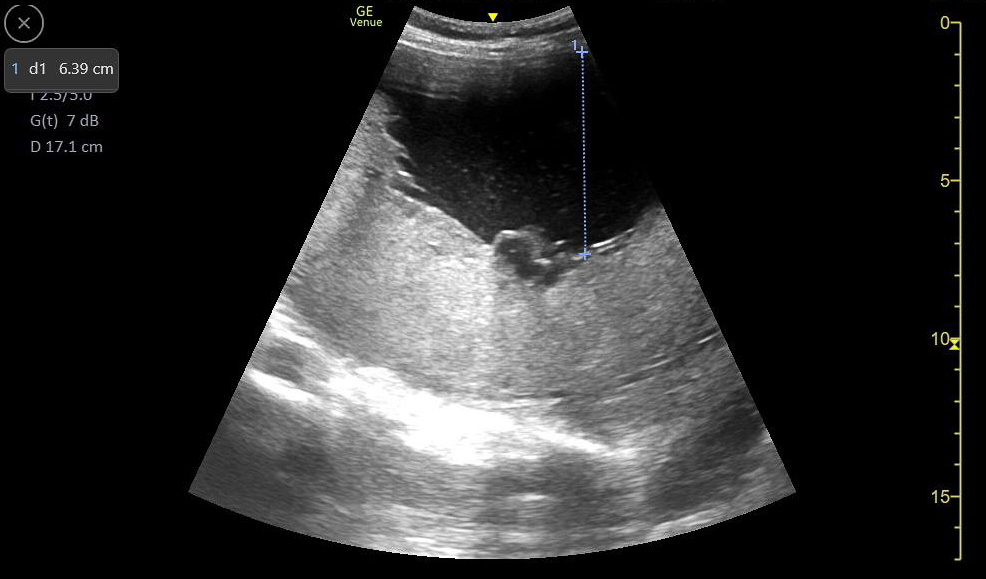
Figure 37. A normal greatest vertical pocket of amniotic fluid. Note the calipers measuring the deepest pocket of amniotic fluid not containing any fetal parts.
Figure 38. No anechoic amniotic fluid is seen surrounding the fetus. Most often due to premature rupture of membranes.
- Polyhydramnios is defined as a greatest vertical pocket of amniotic fluid deeper than 8cm without any fetal parts present.
- If GVP is abnormal, perform a full Amniotic Fluid Index (AFI).
- Measure the single deepest vertical pockets of amniotic fluid in the RUQ, LUQ, LLQ, and RLQ. The total of all quadrants combined should be between 5-25cm.
- Initial GVP instead of AFI is a simpler measure and leads to decreased interventions with no change in patient-oriented outcomes.28
- Fetal Biometry as described in Early Pregnancy chapter
- Biparietal diameter (BPD) and femur length (FL) are traditionally used to date pregnancies in 2nd and 3rd.
- Obstetric sonographic software can use four variables (BPD, FL, Head circumference, and abdominal circumference) to estimate fetal weight.
Gastrointestinal
- Point-of-care ultrasound is useful to diagnose a wide variety of gastrointestinal and surgical pathology, and may be particularly useful in settings where computed tomography (CT) is limited or unavailable.
- Appendicitis29,30
- Small bowel obstruction31
- Intussusception32
- See respective GI chapters for further details
Pneumonia
- Pneumonia is the leading cause of illness and death in children worldwide.33
- Because many cases and mortality occur in RLS where radiography is often unavailable, the WHO developed a case management algorithm based on symptoms, appearance, and vital signs rather than on diagnostic imaging.34
- However, this lacks specificity, with high false positive rate, and may lead to unnecessary antibiotics.35
- A systematic review and meta-analysis of pneumonia in children revealed POCUS to have excellent diagnostic characteristics in resource-rich settings.33
- Sensitivity 96%
- Specificity 93%
- Positive likelihood ratio 15.3
- Negative likelihood ratio 0.06
- These findings were further validated in a recent large study in a busy children’s hospital in a RLS.36
- See lung chapter for further details.
Hydatid Cysts
- A parasitic infectious disease characterized by slow-growing, but often large, cysts that form mostly in the liver, followed by the lungs and spleen.37
- Caused by the cestode Echinoccocus granulosus, with humans ingesting food contaminated by dog feces containing Echinococcus
- Most common in rural pastoral areas as sheep are the definitive host; human infections are incidental.
- Estimated prevalence as high as 6% of population.37
- The parasite hatches in the intestine, travels through the bowel wall to the liver, and then to other organs.
- Growing cysts are often asymptomatic until they produce mass effect or compression of vital structures.
- Though well-visualized on CT and MRI, ultrasound has become the preferred modality for imaging cysts in the liver and spleen.38
- The WHO developed a staging system based on the appearance of the cysts - staging determines treatment course.39
- Stages
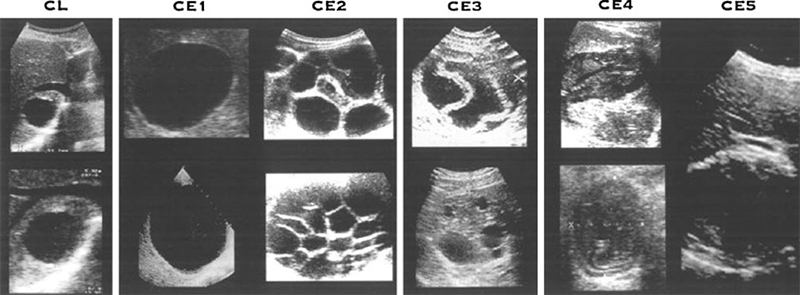
Figure 39. WHO-IWGE standardized classification of types of hydatid cysts. (Reprinted with permission from Elsevier, Acta Tropica Vol. 85, Group WHOIW, International classification of ultrasound images in cystic echinococcosis for application in clinical and field epidemiological settings, 253-261, Copyright 2003)
- CL: spherical fluid collections without a discrete wall, and has no significant internal echoes (not specific for Echinococcus).
- CE1: Dense wall with or without internal echoes.
Figure 40. Multiple CE1 hydatid cysts present in the liver and spleen.
- "Double-wall sign" often seen with high-frequency probe is specific to hydatid cysts and differentiates it from simple cysts.
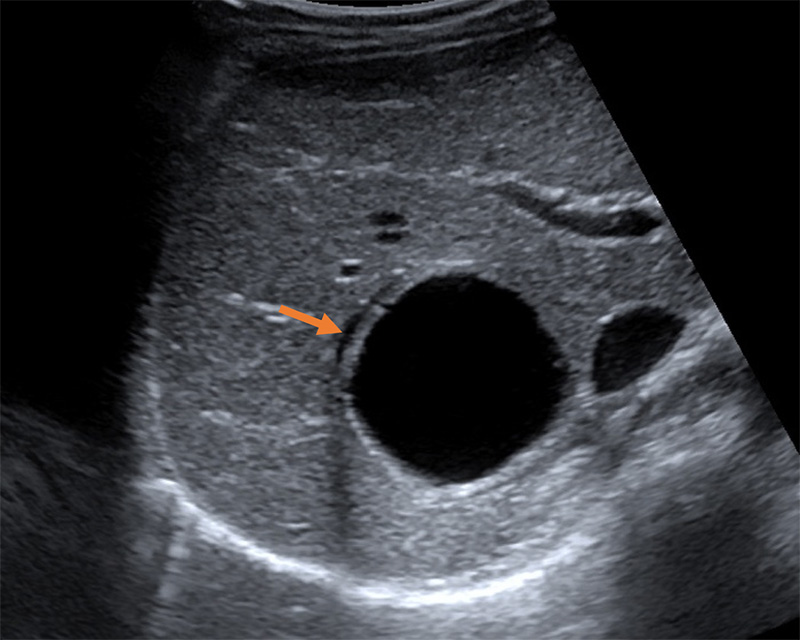
Figure 41. Type CE1 demonstrating the “double wall sign” distinguishing the lesion from other simple cysts. (Liver Imaging Atlas, www.liveratlas.org. Copyright 2010 University of Washington. Image use with permission from University of Washington, accessed 4/14/21)
- CE2: Multiple discrete daughter cysts inside the original cystic capsule, forming a honeycomb pattern.
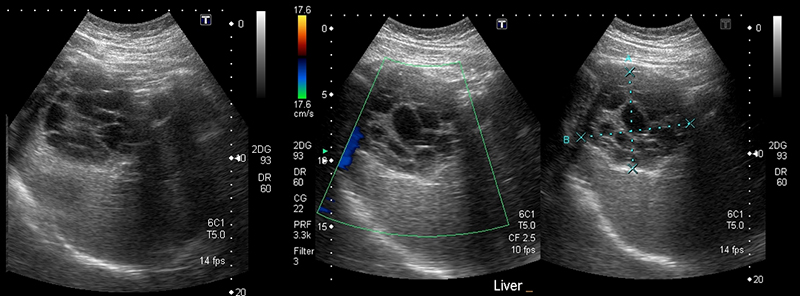
Figure 42. Large single CE2 hydatid cyst with several daughter cysts inside giving characteristic honeycomb appearance. Note the lack of color flow and calipers marking the outer margins in (B). (Case courtesy of Dr Pir Abdul Ahad Aziz Qureshi, radiopaedia.org. From the case rID: 68473, accessed 4/14/21)
- CE3: “transitional” stage as the cyst may either involute and calcify, forming CE4 or CE5 lesions, or reactivate to CE2 cysts.
- CE3a lesions reveal detachment of cystic wall or early collapse (so called “water-lily sign”).
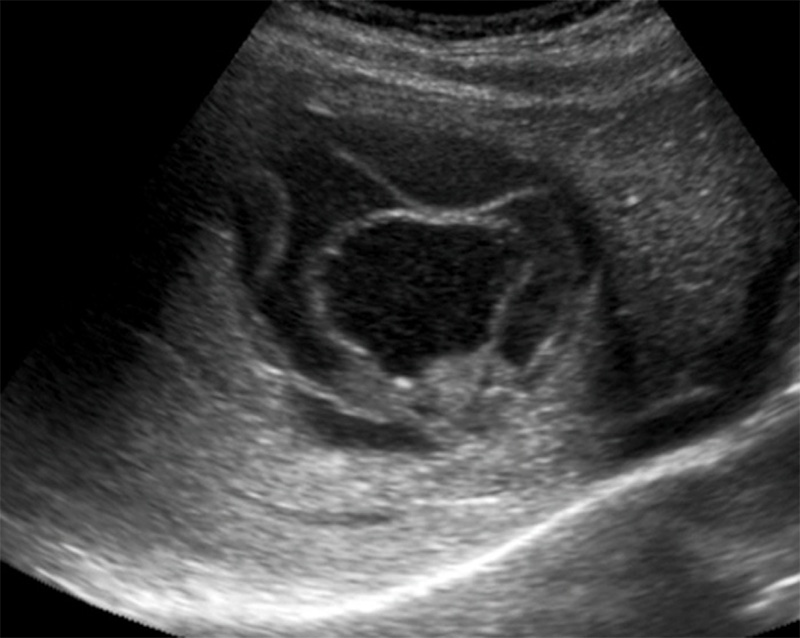
Figure 43. CE3 hydatid cyst with a “water lily sign”, a collapsed endocyst floating within the outer membrane. (Liver Imaging Atlas, www.liveratlas.org. Copyright 2010 University of Washing. Image use with permission from University of Washington, accessed 4/14/21)
- CE3b lesions are mixed cystic and solid lesions
- CE4: heterogenous material instead of fluid-filled cyst cavity, sometimes collapsed membranes
- "Ball of wool" sign: detached endocyst as a hypoechoic folded structure embedded in a hyperechoic matrix
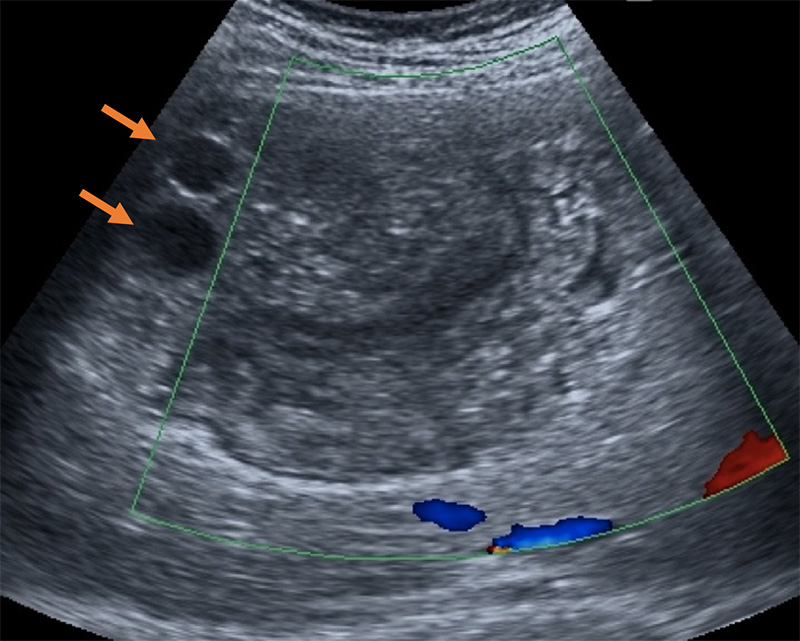
Figure 44. “Ball of wool” sign present in inactive, collapsed endocysts that have formed a surrounding solid matrix. Two cystic daughter cysts are still present in this cyst (arrows). (Liver Imaging Atlas, www.liveratlas.org. Copyright 2010 University of Washington. Image use with permission from University of Washington, accessed 4/14/21)
Figure 45. Type CE4 lesion. (Courtesy of Dr. Doug Wallace, University of Alabama Birmingham Department of Emergency Medicine)
- CE5: thick rim of calcification, involuted, inactive cyst
- Seen only in liver and spleen, takes 5-10yrs to develop
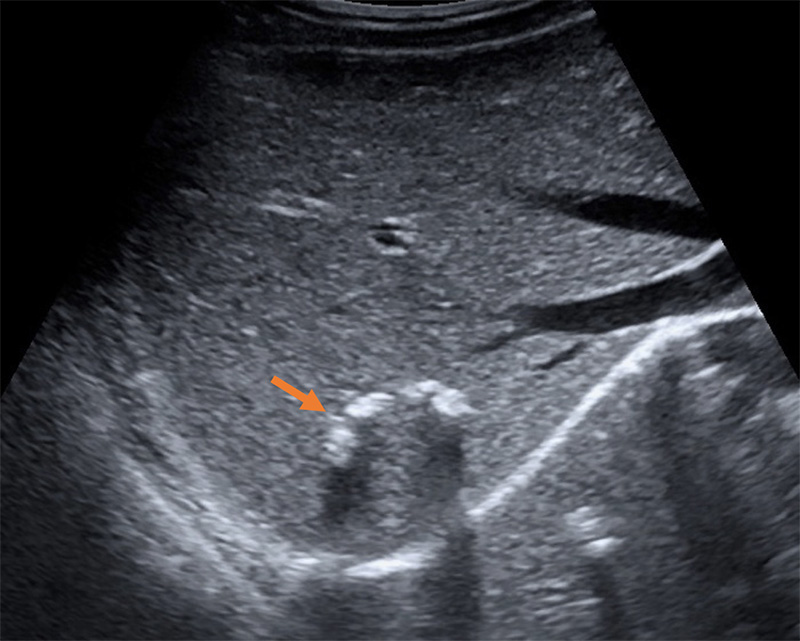
Figure 46. Inactive calcified CE5 cyst (arrow). (Liver Imaging Atlas, www.liveratlas.org. Copyright 2010 University of Washington. Image used with permission from University of Washington, accessed 4/14/21)
Performing the Scan
- Scan the liver with the curvilinear or phased array probe as detailed above, including the left lobe.
- Scan the spleen in both the coronal and axial planes from superior pole to inferior pole. To better visualize the spleen, the probe will likely need to be moved more posteriorly and more superiorly than the corresponding view of the liver. The view may also be improved by rotating the probemarker slightly clockwise to allow scanning between the ribs.
- Subcutaneous cysts can be viewed with a linear transducer.
Treatment
- Management based off sonographic staging, size, location, and potentially influenced by surrounding structures being compressed by mass effect.37
- Medical anti-helminth therapy alone (albendazole with or without praziquantel) when cysts are small (<7cm), in multiple organs, inoperable, or peritoneal.
- Surgery often performed when the cysts are large (>10cm), compressing vital structures, infected, bleeding, or ruptured.
- Rupture of cysts may lead to seeding of other cavities.
- Often combined with medical therapy.
- PAIR – Puncture, Aspiration, Irrigation, and Reaspiration of cyst contents and injection of sporicidal agent.
- Recommended for unilocular abdominal “liquid cysts”, staged CE1 or CE3a, guided by ultrasound.
Schistosomiasis
Overview6
- Affects an estimated 250 million people worldwide, most common in sub-Saharan Africa and Southeast Asia.
- Adult worms live in the gastrointestinal venules (S. mansoni, S. japonicum, S. mekongi) or bladder venous plexus (S. hematobium) and lay eggs which are then deposited in the next distal capillary bed (portal, pulmonary, or bladder).
- Morbidity is driven by the chronic inflammatory response to the eggs, leading to fibrosis of the surrounding hepatic, pulmonary, or genitourinary tissue and portal or pulmonary hypertension or urinary obstruction.
- May lead to significant morbidity including anemia, wasting, stunting, esophageal varices, and squamous cell carcinoma of the bladder.
- Disease burden accumulates with increased exposure and duration of infection, and can eventually lead to life-threatening end-organ damage.
Ultrasound Findings40
- Genitourinary
- Thickening, irregularity, or mass of bladder wall
- Best assessed with a full bladder
- Thickening, irregularity, or mass of bladder wall
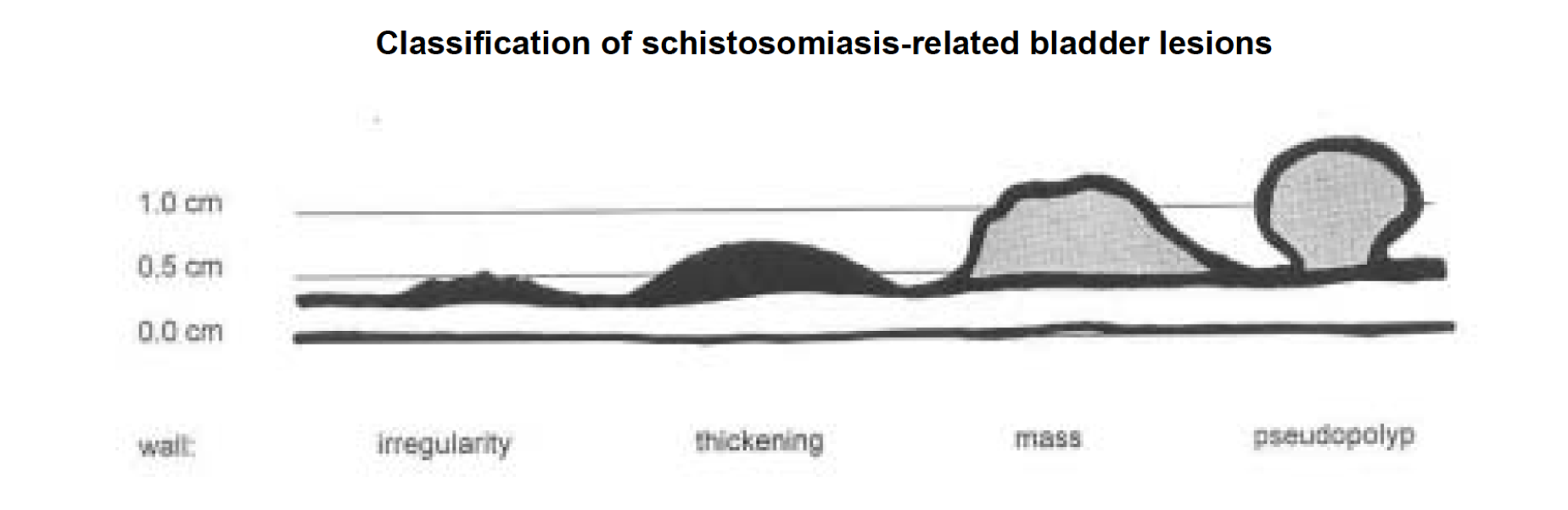
Figure 47. Classification of schistosomiasis-related bladder lesions. (Reproduced with permission, WHO https://apps.who.int/iris/handle/10665/66535)
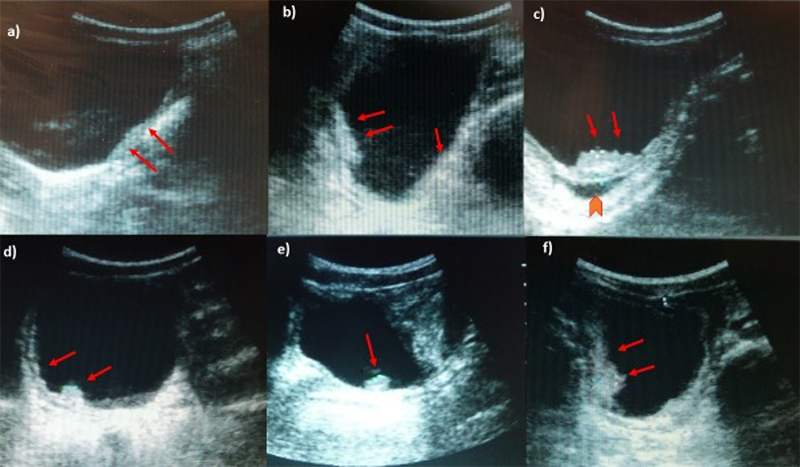
Figure 48. Ultrasonography images of urinary bladder of [School-Aged Children] infected with S. haematobium. a) Thickening of the bladder wall, transverse plane shows thickening of the left lateral wall (arrows); b) Diffuse thickening of the bladder wall more evident in the right posterior wall, echogenic snow in the lumen; c) In oblique longitudinal plane, ultrasound image shows a mass-like lesion in the mucosa layer of the bladder. Block arrow indicates the dilation of the ureter; d) longitudinal plane shows a marked and diffuse thickening of the bladder wall with a mass-like lesion (arrow); e) Mass-like lesion in the absence of a marked and diffuse thickening of the bladder wall; f) Multifocal thickening of the wall, particularly evident on the right and posterior wall. (Reproduced with permission, PLOS https://doi.org/10.1371/journal.pntd.0005400.g003 )
-
- Calcification of bladder wall (pathognomonic but difficult to detect in early stages)
- Hydronephrosis
Figure 49. Mild hydronephrosis seen on long axis view of the kidney. Note the mild dilation of the renal pelvis and calyces and the lack of color Doppler flow over the anechoic (black) dilation of the renal pelvis.
Figure 50. Moderate hydronephrosis seen on long axis view of kidney without a change in the thickness of the renal cortex.
Figure 51. Severe hydronephrosis seen on long axis view of the kidney. Note the severe dilation of the renal pelvis and thinning of the renal cortex, as well as the dilated ureter extending to the right of the image.
-
- Dilatation of ureter
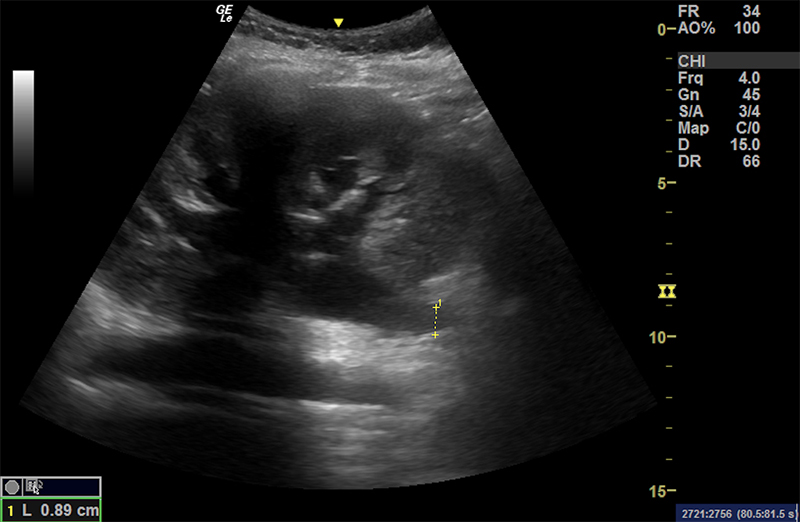
Figure 52. Moderate hydronephrosis with ureteral dilation (extending deep from the renal pelvis).
- Intestinal
- See liver section above for details on how to perform the exam
- Periportal fibrosis – thickening of the hyperechoic borders of the portal vein and branches.
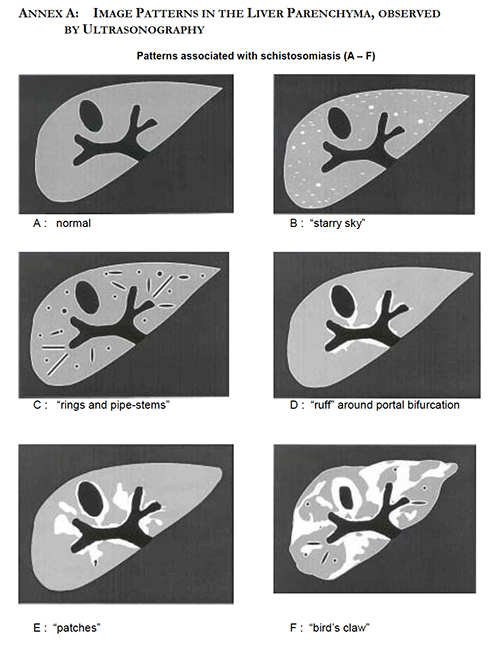
Figure 53. Imaging patterns in the liver parenchyma observed by ultrasonography. Reproduced with permission, WHO https://apps.who.int/iris/handle/10665/66535 )
Figure 54A. Transverse views of the liver revealing moderate periportal fibrosis. Note the thickened hyperechoic walls surrounding the portal vein and branches.
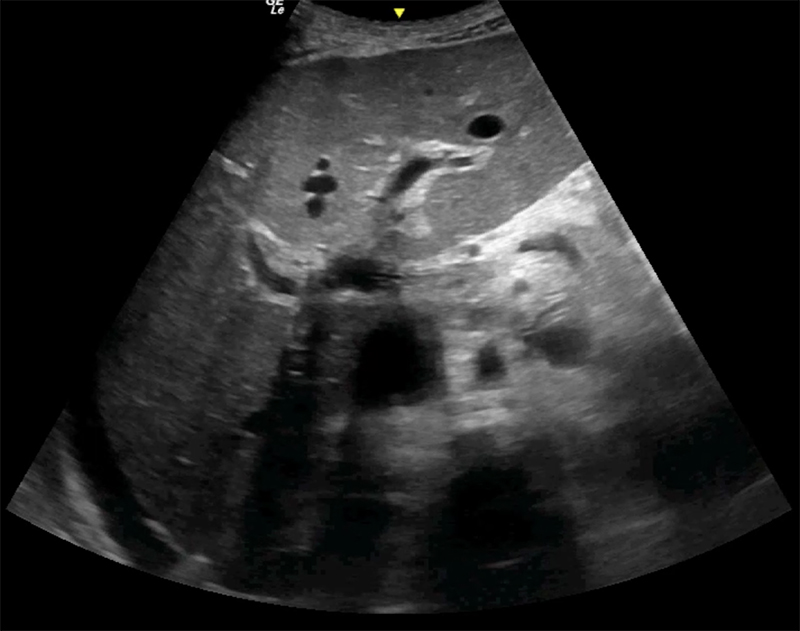
Figure 54B. Transverse views of the liver revealing moderate periportal fibrosis. Note the thickened hyperechoic walls surrounding the portal vein and branches.
-
- Portal hypertension
- Ascites
- Splenomegaly
- Dilated portal vein (>14mm)
- Reversal of portal venous flow by color Doppler
- Gallbladder wall thickening (>3mm)
- May see portal vein thrombosis
- Portal hypertension
- Cardiac
- Chronic pulmonary schistosomiasis is likely the leading cause of pulmonary HTN and RV failure in endemic countries.41
- Detailed description of the diagnosis of pulmonary hypertension by echocardiography is beyond the scope of this work.
- Many point-of-care sonographers are familiar with signs of right heart strain or failure, or increased right heart pressures. Echocardiography may be a useful screening tool.42
- Right atrial dilation (>4cm in any dimension) or increased right atrial area.43
Figure 55. Subxiphoid view revealing severe right atrial dilation, as well as biventricular failure.
-
-
- “D-sign”
- Often combined with RV free wall thickening (>5mm) indicating a chronic process.
- “D-sign”
-
Figure 56. Parasternal short axis view with right ventricular dilation and hypertrophy, as well as septal flattening, creating a chronic “D sign”.
-
-
- Tricuspid regurgitation (moderate to severe)
-
Figure 57. Apical 4 chamber view with severe bilateral atrial dilation, poor ejection fraction, and severe mitral and tricuspid regurgitation.
-
-
- IVC dilation and lack of collapsibility
- Decreased Tricuspid Annular Plane Systolic Excursion (TAPSE), an estimate of right ventricular systolic function obtained by measuring the vertical change in the plane of the lateral tricuspid valve annulus in M-mode in the apical 4 chamber view, to less than 16mm.
-
Utility of Ultrasound4,6
- WHO has widely-used standardized US protocols for both intestinal and urogenital schistosomiasis.40
- Protocols are time-consuming, not initially designed for POCUS use and some authors recommend moving towards a more qualitative, simplified approach. 4,44,45
- The authors in general do not perform the entire protocol in the acute setting, but look for qualitative findings indicating that complications from chronic schistosomiasis may be related to the acute presentation.
- Simplified protocols have shown high sensitivity and specificity even with novice sonographers.46
- Transverse and longitudinal views of the bladder – assess for bladder wall lesions, masses, pseudopolyps, or calcifications.
- Post-void residual view of bladder.
- Transverse and coronal views of bilateral kidneys – assess for hydronephrosis and hydroureter.
- Parasagittal right anterior axillary line liver view – assess for hepatomegaly and ascites.
- Coronal view of liver between right anterior and mid-axillary lines – use color Doppler to assess direction of portal venous flow.
- Substernal transverse view of liver – assess left lobe of liver.
- US can readily identify signs of complications such as portal and pulmonary hypertension and urinary obstruction.
- This application may be of the greatest use to acute care or emergency clinicians in high-risk settings.
- US may be used to grade the severity of known disease.
- US may be used to evaluate the response to treatment with praziquantel.
- Improvement or resolution of mild periportal fibrosis and urinary lesions can be followed by ultrasound in patients with less severe infection.47-49
- Because severe disease is less likely to result in improvement after treatment, US may be useful for screening at-risk patients prior to onset of irreversible disease.
- Limited evidence exists for the use of point-of-care echocardiography in the management of schistosomiasis-associated pulmonary hypertension.42
- Signs of RV failure or dilation in an undifferentiated dyspneic patient in a Schistosoma-endemic region may aid in diagnosis.
- Simplified protocols have shown high sensitivity and specificity even with novice sonographers.46
Dengue
Overview
- Tropical viral disease transmitted by Aedes spp. mosquitoes that infects an estimated 390 million per year, of which 96 million are clinically apparent.50
- Characterized by headache/retro-orbital pain, profound muscle and bone aches, fever, and rash.51
- Febrile phase of 2-7 days
- Critical or defervescent phase lasting 1-2 days in which patients will either improve or develop increased vascular permeability and plasma leakage.
- May lead to hemoconcentration and potentially shock.
- Some also develop profound thrombocytopenia, DIC, or other hemorrhagic complications.
- Recovery phase in which patients resorb extravascular fluids over 2-3 days
Disease Severity Classifications51
- Dengue Fever (DF)
- Presumptive (or confirmed) diagnosis + Fever + 2 or more of:
- Nausea/Vomiting
- Rash
- Headache/eye pain
- Arthralgias/myalgias
- Leukopenia
- (+) tourniquet test (a clinical marker of thrombocytopenia)
- Presumptive (or confirmed) diagnosis + Fever + 2 or more of:
- Dengue Fever with Warning Signs (DF WS)
- Above findings plus any
- Abdominal pain/tenderness
- Persistent vomiting
- Clinical fluid accumulation (ascites, pleural or pericardial effusion)
- Mucosal bleeding
- Lethargy
- Hepatomegaly
- Increase in hematocrit with decrease in platelet count
- Above findings plus any
- Severe Dengue Fever (SDF)
- Dengue infection with >1 of
- Severe plasma leakage causing shock or fluid accumulation with respiratory distress
- Severe bleeding
- Severe end organ damage
- Dengue infection with >1 of
Ultrasound Findings and Application
- A recent meta-analysis found significant heterogeneity among studies evaluating POCUS in DF or SDF (mostly regarding timing of ultrasound) preventing definitive conclusions about its predictive and diagnostic value.52
- There are several ultrasound findings that may prove particularly useful in an endemic area where confirmatory testing such as NS1 or serology is unavailable.52,53
- Plasma leakage and third spacing
- Ascites
- Pleural effusions
- Pericardial effusion
- All tend to develop during “critical”
- Effusions and ascites are markers of more severe disease.54,55
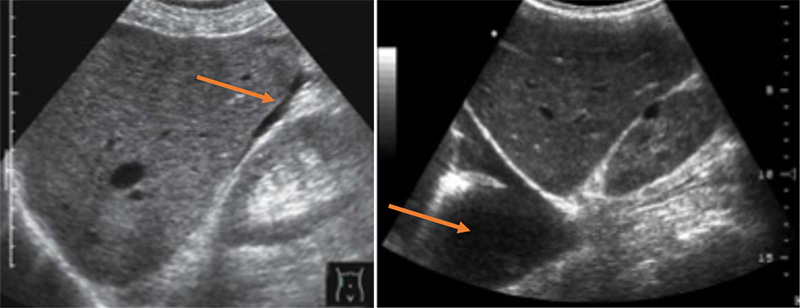
Figures 58A & B. Fluid third-spacing in severe dengue demonstrated via views of free fluid (arrow) in Morrison’s pouch (A), the pleural space (arrow) (B). (Courtesy of Dr. Elijah Katz, Medicines Sans Frontieres)
Figure 58C. Fluid third-spacing in severe dengue demonstrated via view of free fluid (arrow) in the pericardium.
- Gallbladder wall thickening (GBWT)
- Some studies have shown that GBWT may follow distinct progressive patterns that seem to correlate with disease severity.55
- Parmar et al. found GBWT in 96.5% of SDF and only 28% of DF.53
- They concluded that GBWT had a high positive predictive value in patients in patients in whom DF was clinically suspected.
- Negative predictive value and sensitivity were too low to be adequate screening tools.
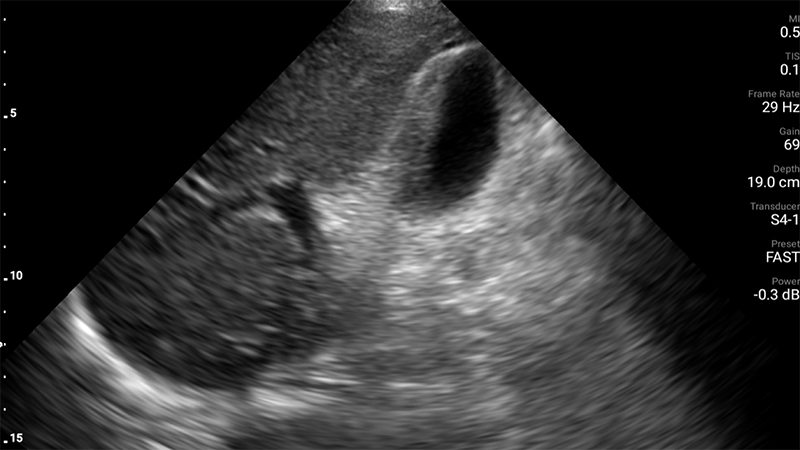
Figure 59. Asymmetric gallbladder wall thickening (arrow). Note that biliary sludge (if present) would be on the opposite, dependent side of the gallbladder lumen. (Courtesy of Dr. Sashita Shah, University of Washington, Seattle, WA, USA)
Figure 60. Gallbladder wall thickening in a pediatric case of severe dengue that had no other findings of third spacing.
- Hepatomegaly, splenomegaly, and mesenteric lymphadenopathy also seen with severe dengue.
- Pleural effusions tend to occur more frequently than ascites, and gallbladder wall thickening and hepatomegaly are more strongly associated with severe dengue.56
- Signs of GBWT or plasma leakage could indicate impending deterioration into SDF, or need for higher level of care or intervention, though further studies are needed.6
Performing the Exam
- The FASH exam, as described below, obtains all the views necessary to assess for pleural and pericardial effusions, ascites, mesenteric lymphadenopathy, as well as hepatosplenomegaly.
- Gallbladder assessment is in a similar fashion to a typical gallbladder ultrasound.
FASH – Focused Assessment with Sonography in HIV-Associated Tuberculosis
- Tuberculosis (TB) is the single leading cause of infectious death and a top ten overall cause of death worldwide.57
- There is substantial overlap in areas of highest prevalence of TB and HIV with many of these locations having significantly limited diagnostic resources.
- Coinfection of TB and HIV increases risk of disseminated or extrapulmonary TB (EPTB) by 20-30x.58
- Diagnosis of EPTB may be more difficult given limited diagnostic resources in endemic settings and by limited immune response in persons living with HIV leading to lack of chest x-ray findings or PPD response.59
- The FASH exam was developed to aid in diagnosis of EPTB in HIV+ patients in areas of high TB prevalence.
- One or more of the following findings increases the likelihood of EPTB in patients living with HIV:
- Pericardial effusion
- Ascites
- Periaortic lymphadenopathy
- Hepatic or splenic microabscesses
- A positive FASH has lead to changes in management (most often the initiation of TB therapy) in up to 72% of patients.59
- 2 or more findings on FASH exam was 91% specific for EPTB in South Africa.60
- See FASH chapter for further details.
- One or more of the following findings increases the likelihood of EPTB in patients living with HIV:
LOGISTICS
To scan effectively in a RLS, several elements must be in place:
- A small, mobile, relatively rugged ultrasound machine
- ACEP’s Global Emergency ultrasound subcommittee has produced a helpful guide.
- Gel
- Can be transported, purchased in many countries, or home-made
- Multiple authors have performed comparisons of commercial and homemade recipes in RLS.61,62
- Disinfection wipes or protocol
- Power or batteries
- Many machines must be connected to main power or a generator to scan effectively.
- If main power is not available, solar powered generators may be used to power machines or recharge batteries.
- If battery powered, consider options to bring extra batteries or back up charging solutions.
- Towels or wipes for patient clean up
- Privacy
- Considerations must be made for patient privacy while scanning, especially in more traditional or conservative cultures.
- This could include a separate room, secluded outdoor station, or privacy screens.
- Knowledge of POCUS
- Technical skills in image acquisition and interpretation.
- Appropriate integration of POCUS into clinical decision-making.
- Knowledge of tropical infectious diseases
- Epidemiology - Which tropical diseases is the patient at risk for?
- Pathophysiology - How does the particular tropical infection affect the patient, and are those effects detectable on ultrasound?
- Management – How do the ultrasound findings affect management of this patient?
- Realistic assessment of resources
- What other diagnostic modalities are available? Is POCUS the best (or only) strategy?
- POCUS often does not reveal a definitive diagnosis, but can offer significant data for the next step acutely.
- What can be done with the diagnosis? What interventions are available in the particular context?
- What other diagnostic modalities are available? Is POCUS the best (or only) strategy?
- More resources discussing the logistics of establishing a self-sufficient POCUS program.
- Henwood et al.63
- Global Point-of-care Ultrasound: A Technical Guide
References
Introduction
- Stolz LA, Muruganandan KM, Bisanzo MC, et al. Point-of-care ultrasound education for non-physician clinicians in a resource-limited emergency department. Trop Med Int Health. 2015;20(8):1067-72.
- Sippel S, Muruganandan K, Levine A et al. Review article: use of ultrasound in the developing world. Int J Emerg Med. 2011;4:72
- Accreditation Council for Graduate Medical Education. (2016) Program requirements for graduate medical education in emergency medicine. Revised common program requirements. https://www.acgme.org/Portals/0/PFAssets/ProgramRequirements/CPRs_2017-07-01.pdf Accessed November 27, 2020.
- Belard S, Tamarozzi F, Bustinduy AL, et al. Point-of-care ultrasound assessment of tropical infectious diseases – a review of applications and perspectives. Am J Trop Med. 2016;94(1): 8-21.
- Brunetti E, Heller T, Richter J, et al. Application of ultrasonography in the diagnosis of infectious disease in resource-limited settings. Curr Infect Disease Report. 2016;18:6
- Kaminstein D, Kuhn WT, Huang D, et al. Perspectives on point-of-care ultrasound use in pediatric tropical infectious disease. Clin Pediatric Emerg Med. 2019;20(2):128-140.
Rheumatic Heart Disease
- Watkins DA, Johnson CO, Colquhoun SM, et al. Global, Regional, and National Burden of Rheumatic Heart Disease, 1990-2015. N Engl J Med. 2017;377(8):713-722.
- Gewitz MH, Baltimore RS, Tani LY, et al. American Heart Association Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease of the Council on Cardiovascular Disease in the Young. Revision of the Jones Criteria for the diagnosis of acute rheumatic fever in the era of Doppler echocardiography: a scientific statement from the American Heart Association. Circulation. 2015 May 19;131(20):1806-18 Epub 2015 Apr 23. Erratum in: Circulation. 2020 Jul 28;142(4):e65. PMID: 25908771.
- Longenecker CT. Echo Screening for Rheumatic Heart Disease. Circ Cardiovasc Imaging. 2019 Feb;12(2):e008818.
- Mirabel M, Bacquelin R, Fafflet M, et al. Screening for rheumatic heart disease: evaluation of a focused cardiac ultrasound approach. Circ Cardiovasc Imaging 2015;8:e002324.
- Nunes MCP, Sable C, Nascimento BR, et al. Simplified Echocardiography Screening Criteria for Diagnosing and Predicting Progression of Latent Rheumatic Heart Disease. Circ Cardiovasc Imaging. 2019 Feb;12(2):e007928.
Liver Disease
- Asrani SK, Devarbhavi H, Eaton J, Kamath PS. Burden of liver diseases in the world. J Hepatol. 2019;70(1):151-171.
- Ultrasoundpaedia Pty Limited. Ultrasound of the Liver. Normal Liver Ultrasound – How To.https://www.ultrasoundpaedia.com/normal-liver2/. Accessed November 9, 2020.
- Gelbard RB, Mahendran A, Hardy MA.Chapter 6. Abdominal Ultrasound. In: Schrope B. Surgical and Interventional Ultrasound. McGraw-Hill; Accessed November 9, 2020. https://accesssurgery-mhmedical-com.ezproxy3.lhl.uab.edu/content.aspx?bookid=698§ionid=45954786.
- Heller T.Focused Assessment with Sonography for HIV/TB – a Practical Manual. (TALC – Teaching Aids at Low Cost, ed.). Munich: Copy and Print; 2013.
- Dietrich CF, Serra C, Jedrzejczyk M, Dietrich CF. Ultrasound of the Liver. In:EFSUMB – European Course Book. Bologna: University of Bologna; 2010:1-90.
- Hertzberg BS and Middleton WD. Chapter 3 Liver. In: Ultrasound: The Requisites. 3rd Philadelphia, PA.: Elsevier, 2016.
- Kurland JE and Brann OS. Pyogenic and Amebic Liver Abscesses. Curr Gastroenterol Rep. 2004;6(4)273-9.
Obstetrics
- McClure EM, Goldenburg RL, Bann CM. Maternal mortality, stillbirth, and measures of obstetric care in developing and developed countries. Int J Gynaecol Obstet. 2007;96:139-146.
- Abuhamad A, Chaoui R, Jeanty P, and Paladini D. Stepwise standardized approach to the basic obstetric ultrasound examination in the second and third trimester. In: Abuhamad A, ed.. Ultrasound in Obstetrics and Gynecology: A Practical Approach. 2014. https://www.evms.edu/obstetrics_and_gynecology/ultrasound_in_obstetrics_and_gynecology/ Accessed 14 November 2020.
- Goldenberg RL, Nathan RO, Swanson D, et al. Routine antenatal ultrasound in low- and middle-income countries: first look – a cluster randomized trial. Br J Obstet Gynaecol 2018;125(12):1591-1599.
- Shah SP, Epino H, Bukhman G, et al. Impact of the introduction of ultrasound devices in a limited resource setting: rural Rwanda 2008. BMC Int Health Hum Rights. 2009;9:4.
- Becker DM, Tafoya CA, Becker SL, Kruger GH, Tafoya MJ et al. The use of portable ultrasound devices in low- and middle-income countries: a systematic review of the literature. Trop Med Int Health. 2016;21:294-311.
- Kimberly HH, Murray A, Mennike M, Liteplo A, Lew J, et al. Focused maternal ultrasound by midwives in rural Zambia. Ultrasound Med Biol. 2010;36:1267-72.
- Bellusi F, Ghi T, Youssef A, et al. The use of intrapartum ultrasound to diagnose malpositions and cephalic malpresentations. Am J Obstet Gynecol. 2017;217(6):633-641
- Abuhamad A, Chaoui R, Jeanty P, and Paladini D. Ultrasound evaluation of twin gestation. In: Abuhamad A, ed. Ultrasound in Obstetrics and Gynecology: A Practical Approach. 2014. https://www.evms.edu/obstetrics_and_gynecology/ultrasound_in_obstetrics_and_gynecology/ Accessed 14 November 2020.
- Abuhamad A, Chaoui R, Jeanty P, and Paladini D. Placental Abnormalities. In: Abuhamad A, ed. Ultrasound in Obstetrics and Gynecology: A Practical Approach. 2014. https://www.evms.edu/obstetrics_and_gynecology/ultrasound_in_obstetrics_and_gynecology/ Accessed 14 November 2020.
- Nabhann AF, Abdelmoula YA. Amniotic fluid index versus single deepest vertical pocket as a screening test for preventing adverse pregnancy outcome. Cochrane Database Syst Rev. 2008(3).
Gastrointestinal
- Lee HL and Seong JY. Diagnostic performance of emergency physician-performed point-of-care ultrasonography for acute appendicitis: A meta-analysis. Am J Emerg Med. 2019;37(4):696-705.
- Gungor F, Kilic T, Akyol KC, et al. Diagnostic value and effect of bedside ultrasound in acute appendicitis in the emergency department. Acad Emerg Med. 2017;24:578-86.
- Gottlieb M, Peksa GD, Pandurangadu AV, et al. Utilization of ultrasound for the evaluation of small bowel obstruction: a systematic review and meta-analysis. Am J Emerg Med. 2018;36:234-42.
- Trigylidas TE, Hegenbarth MA, Patel L, et al. Pediatric emergency medicine point-of-care ultrasound for the diagnosis of intussusception. J Emerg Med. 2019;57(3)367-74.
Pneumonia
- Pereda MA, Chavez MA, Hooper-Miele CC, et al. Lung ultrasound for the diagnosis of pneumonia in children: a meta-analysis. Pediatrics. 2015;135(4):714-22.
- World Health Organization. Technical basis for the WHO recommendations on the management of pneumonia in children at first level health facilities. Geneva: World Health Organization, 1991. (WHO/ARI/91.20).
- Hazir, T, Nisar YB, Abbasi S, et al. Comparison of oral amoxicillin with placebo for treatment of World Health Organization-defined nonsevere pneumonia in children aged 2-59 months: a multicenter, double-blind, randomized, placebo-controlled trial in Pakistan. Clin Infect Dis. 2011;52:293-300.
- Ellington LE, Gilman RH, Chavez MA, et al. Lung ultrasound as a diagnostic tool for radiographically confirmed pneumonia in low resource settings. Respir Med. 2017;128:57-64.
Hydatid Cysts
- Shantz PM, Kern P, Brunetti E. Chapter 120 – In: Guerrant RL, Walker DH, Weller PF. Tropical infectious disease: principles, pathogens, and practice. W.B. Saunders, 2011. 824-838. https://doi.org/10.1016/B978-0-7020-3935-5.00120-8.
- Brunetti E, Tamarozzi F, Macpherson C, Filice C, Piontek MS, Kabaalioglu A, Dong Y, Atkinson N, Richter J, Schreiber-Dietrich D, Dietrich CF. Ultrasound and Cystic Echinococcosis. Ultrasound Int Open. 2018 Sep;4(3):E70-E78.
- Group WHOIW. International classification of ultrasound images in cystic echinococcosis for application in clinical and field epidemiological settings. Acta Trop. 2003;85:253-261.
Schistosomiasis
- Richter J, Hatz C, Campagne G, et al. Ultrasound in schistosomiasis: a practical guide to the standard use of schistosomiasis-related morbidity. Second international workshop. October 22-26, 1996, Niamey, Niger. Revised and updated. World Health Organization. Available at https://apps.who.int.iris.handle/10665/66535
- Papamatheakis DG, Mocumbi AOH, Kim NH, and Mandel J. Schistosomiasis-associated pulmonary hypertension. Pulm Circ. 2014;4(4):596-611.
- Nunes MCP, Guimaraes Jr MH, Diamantino AC, Gelape CL, Ferrari TCA. Cardiac manifestations of parasitic diseases. 2017;103:651-658.
- Carvalho VT, Barbosa MM, Nunes MCP, et al. Early right cardiac dysfunction in patients with Schistosomiasis mansoni. 2011;28(3):261-7.
- El Sheich T, Holtfreter MC, Ekamp H, et al. The WHO ultrasonography protocol for assessing hepatic morbidity due to Schistosoma mansoni. Acceptance and evolution over 12 years. Parasitol Res. 2014;113:3915-25.
- Akpata R, Neumayr A, Holtfreter MC, et al. The WHO ultrasonography protocol for assessing morbidity and mortality due to Schistosoma hematobium. Acceptance and evolution over 14 years. Systematic Review. Parasitol Res. 2015;14:1279-1289.
- Barsky M, Kushner L, Ansbro M, et al. A feasibility study to determine if minimally trained medical students can identify markers of chronic parasitic infection using bedside ultrasound in rural Tanzania. World J Emerg Med. 2015;6:293-8.
- Barda B, Coulibaly JT, Hatz C, Keiser J. Ultrasonographic evaluation of urinary tract morbidity in school-aged and preschool-aged children infected with Schistosoma heaematobium and its evolution after praziquantel treatment: A randomized controlled trial. PLoS Neg Trop Dis 2017;11(2): e0005400.
- Berhe N, Myrvang B, Gundersen SG. Reversibility of schistosomal periportal thickening/fibrosis after praziquantel therapy: a twenty-six month follow-up study in Ethiopia. Trop Med Hyg. 2008;78:228-34.
- Bocanegra C, Pintar Z, Mendioroz J, et al. Ultrasound evaluation of pediatric urinary schistosomiasis after treatment with praziquantel in a highly endemic area. Trop Med Hyg. 2018;99:1011-7
Dengue
- Bhatt S, Gething PW, Brady OJ, et al. The global distribution and burden of dengue. 2013;496(7446):504-7.
- World Health Organization. Dengue: Guidelines for diagnosis, treatment, prevention, and control. Geneva: WHO; 2009.
- Low GKK, Looi SY, Yong MH, et al. Predictive and diagnostic accuracy of ultrasonography in differentiating severe dengue from nonsevere dengue. J Vector Borne Dis. 2018;55:79-88.
- Parmar J, Mohan C, Kumar CP, et al. Ultrasound is not a useful screening tool for dengue fever. Pol J Radiol. 2017; 82;693-700.
- Bharath KR, Laksmana RR, Veerappa BG, et al. Ultrasonography as a tool in predicting the severity of dengue fever in children – a useful aid in a developing country. Pediatr Radiol. 2013;43:971-977.
- Parmar JP, Mohan C, Vora M. Patterns of Gall Bladder Wall Thickening in Dengue Fever: A Mirror of the Severity of Disease. Ultrasound Int Open. 2017;3(2):E76-E81.
- Tavares MA Joao GAP, Bastros MS, et al. Clinical relevance of gallbladder wall thickening for dengue severity: A cross-sectional study. PLoS One. 2019;14(8):e0218939.
FASH
- World Health Organization. Tuberculosis global facts. Geneva: WHO; 2019.
- World Health Organization. Tuberculosis fact sheet No. 104. Geneva: WHO; 2016.
- Heller T, Mtemang’ombe EA, Huson MAM, et al. Ultrasound for patients in high HIV/tuberculosis prevalence settings: a needs assessment and review of focused applications for sub-Saharan Africa. Int J Infect Dis. 2017;56:229-236.
- Van Hoving DJ, Kenge AP, Maartens G, Meintjes G. Point-of-care ultrasound predictors for the diagnosis of HIV-positive patients presenting to an emergency center. J Acquir Immune Defic Syndr. 2020;83(4):415-423.
Logistics
- Cherukuri AR, Lane L, Guy D, et al. Shake no back: a homemade ultrasound gel recipe for low-resource settings. J Ultrasound Med. 2019;38:1069-1073.
- Vinograd AM, Fasina A, Dean AJ et al. Evaluation of noncommercial ultrasound gels for use in resource-limited settings. J Ultrasound Med. 2019;38:371-377.
- Henwood PC, Mackenzie DC, Rempell JS et al. A practical guide to self-sustaining point-of-care ultrasound education programs in resource-limited settings. Ann Emerg Med. 2014;64:277-285.